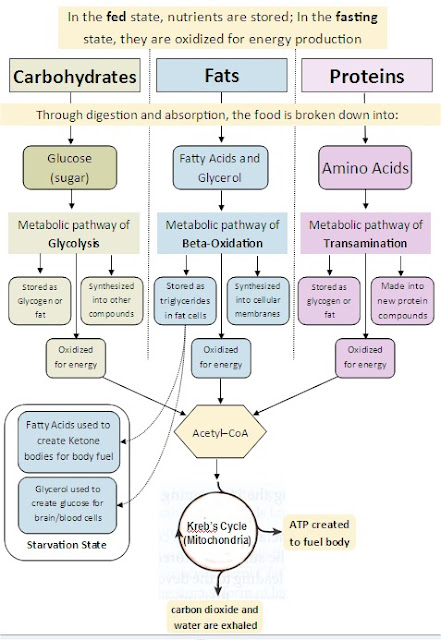
Homeostasis is very important for life. Many factors need to stay within certain parameters, or an organism will die. Temperature, pH, blood pressure, concentrations of nutrients and wastes, etc. all have levels that are optimal and necessary. Staying within these parameters is homeostasis.
In order to maintain the proper levels, many negative feedback loops are utilized. Something triggers the release of something, which then inhibits the release of more. The classic example is a thermostat. When the temperature drops down below a set level, the furnace kicks on and puts out heat. Once the temperature has reached the proper level, that shuts the furnace off. If it didn't have the negative feedback, the furnace would never shut off.
In the thermostat example, the factor would be temperature, the sensor is the thermometer, the integrating center is the thermostat, and the effector is the furnace (the thing that does the action to make the change to get you back to homeostasis). The looped red arrow indicates that when the effector (furnace) does its job, that has an effect on the factor (temperature), which then causes it to stop the loop and turn the furnace off once it's reached the right level.
Here is an example in the body, with blood pressure.
When blood pressure (factor) increases, it is sensed in the baroreceptors in the carotid arteries (sensor), which sends the signal to the brain (integrating center), which sends signals to the heart (effector) to decrease the heart rate. This will lower the blood pressure so that the stimulus is not continued, and the loop stops.
Just FYI, there are some positive feedback loops in the body as well, which do
not contribute to homeostasis, but have different functions. Eventually the stimulus resolves itself so the positive feedback goes away. An example is birth. Stretching of the cervix releases oxytocin which increases contractions, which makes cervix stretch more, which increases oxytocin, and so on, until the baby is born and that stimulus is resolved.